Online Database of Chemicals from Around the World
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Nantong C&R Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chenrunchem.com | |||
 | +86 (513) 8874-3581 +86 15358700783 | |||
 | +86 (513) 8297-1198 | |||
 | chenrunchem@sina.com 909600256@qq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Guangzhou Jun Xin Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jxinchem.com | |||
 | +86 (020) 8256-9208 | |||
 | +86 (020) 2826-9100 | |||
 | ivyyoung@gzjxchem.com | |||
 | QQ Chat | |||
 | WeChat: gzjx18122041046 | |||
 | WhatsApp:+8618122041046 | |||
| Chemical distributor since 2007 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | Catalysts and additives >> Polyethylene glycol derivative |
|---|---|
| Name | Polyethylene glycol mono(2-ethylhexyl) ether |
| Synonyms | Berol 840; Blaunon EH 2; Blaunon EH 4; Blaunon EH 6; EH 6; Ecosurf EH 6; Ethoxylated 2-ethylhexanol; Imbentin OA 100; Koremul OT 5; Newcol 1004; Newcol 1006; Newcol 1008; Newcol 1008GC; Newcol 1020; Nissan Nonion EH 202; Nissan Nonion EH 203; Nonion EH 202; Nonion EH 203; PEH 6 |
| Molecular Structure | 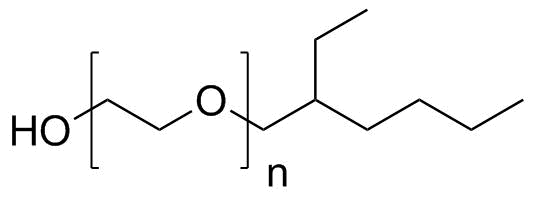 |
| Molecular Formula | x(C2H4O).C8H18O |
| CAS Registry Number | 26468-86-0 |
| EC Number | 607-943-2 |
| SMILES | CCCCC(CC)COCCO |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H412 Details | ||||||||||||||||||||||||
| Safety Statements | P264-P264+P265-P273-P280-P302+P352-P305+P351+P338-P321-P332+P317-P337+P317-P362+P364-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
|
Polyethylene glycol mono(2-ethylhexyl) ether is a nonionic surfactant composed of a polyethylene glycol (PEG) chain terminated at one end by a hydrophilic hydroxyl group and at the other end by a hydrophobic 2-ethylhexyl ether group. Its structure gives it amphiphilic properties, allowing it to interact with both aqueous and nonpolar phases, which is the basis for its function as an emulsifier, wetting agent, and solubilizer. The molecule consists of a polyethylene glycol backbone, which is a repeating unit of ethylene oxide (–CH2CH2O–)n, where the degree of polymerization (n) determines its chain length and hydrophilicity. One terminal hydroxyl group is modified by etherification with a branched 2-ethylhexyl group, a hydrophobic eight-carbon alkyl chain with branching that increases steric bulk and influences surfactant behavior. The opposite end of the PEG chain typically remains as a hydroxyl group, contributing to water solubility. This class of ethoxylated alcohol derivatives emerged from developments in industrial surfactant chemistry during the mid-20th century, when ethylene oxide polymerization techniques were widely adopted to produce nonionic surfactants with tunable properties. By adjusting the length of the polyethylene glycol chain and the nature of the hydrophobic alkyl group, chemists were able to design surfactants with specific hydrophilic–lipophilic balance (HLB) values suitable for different applications. Polyethylene glycol mono(2-ethylhexyl) ether is typically produced through the reaction of 2-ethylhexanol with ethylene oxide under controlled catalytic conditions. The ethoxylation process adds repeating ethylene oxide units to the alcohol, forming a PEG chain of defined length. The resulting product is a mixture of oligomers with varying chain lengths, which is common in industrial ethoxylated surfactants. In aqueous systems, the compound exhibits surface-active behavior due to its amphiphilic structure. The hydrophobic 2-ethylhexyl group tends to associate with nonpolar substances such as oils and organic compounds, while the hydrophilic PEG chain interacts with water molecules through hydrogen bonding. This dual affinity allows the molecule to reduce surface tension and stabilize emulsions by positioning itself at the interface between oil and water phases. As a nonionic surfactant, polyethylene glycol mono(2-ethylhexyl) ether is less sensitive to changes in pH and ionic strength compared to ionic surfactants. This makes it useful in a wide range of formulations where stability under varying chemical conditions is required. It is commonly used in industrial and technical applications such as emulsification, dispersion of hydrophobic materials, and formulation of cleaning agents, coatings, and agrochemical products. The branched nature of the 2-ethylhexyl group contributes to improved fluidity and reduced crystallization compared to linear alkyl chains. This structural feature enhances the compound’s performance in liquid formulations by maintaining solubility and preventing phase separation under a range of temperatures. Polyethylene glycol-based surfactants, including mono(2-ethylhexyl) ether derivatives, are also studied for their role in modifying interfacial properties in chemical processes. The PEG segment provides hydration and steric stabilization, while the hydrophobic moiety anchors the molecule at interfaces. This combination is particularly useful in stabilizing emulsions and dispersions in complex formulations. Overall, polyethylene glycol mono(2-ethylhexyl) ether is a nonionic amphiphilic surfactant derived from ethoxylated alcohol chemistry. Its structure, combining a hydrophilic polyethylene glycol chain with a branched hydrophobic alkyl ether group, gives it broad utility in industrial formulations requiring emulsification, solubilization, and surface modification. References 2025. Exploiting metal-free atom transfer radical polymerization for fabrication of styrene-maleic acid polymer-brush grafting chromatographic stationary phase and its separation performances for phospholipids. Analytical and Bioanalytical Chemistry. DOI: 10.1007/s00216-025-06053-1 2022. Influence of oak chips addition on the physicochemical properties of beer. European Food Research and Technology. DOI: 10.1007/s00217-022-04143-3 2021. Analysis of Glass Transition Temperature Shifts Induced by Internal and External Plasticizers in (Meth)acrylate Polymer Coatings with Molecular Simulations. Proceedings of the 2nd International Conference on Advanced Surface Enhancement (INCASE 2021). DOI: 10.1007/978-981-16-5763-4_55 |
| Market Analysis Reports |