Online Database of Chemicals from Around the World
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Guangzhou Jun Xin Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jxinchem.com | |||
 | +86 (020) 8256-9208 | |||
 | +86 (020) 2826-9100 | |||
 | ivyyoung@gzjxchem.com | |||
 | QQ Chat | |||
 | WeChat: gzjx18122041046 | |||
 | WhatsApp:+8618122041046 | |||
| Chemical distributor since 2007 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | Catalysts and additives >> Polymer |
|---|---|
| Name | Isotridecanol ethoxylate |
| Synonyms | PEG isotridecyl ether; Poly(ethylene glycol) mono(isotridecanol) ether; Polyethylene glycol isotridecyl ether; Polyethylene glycol isotridecyl monoethe |
| Molecular Structure | 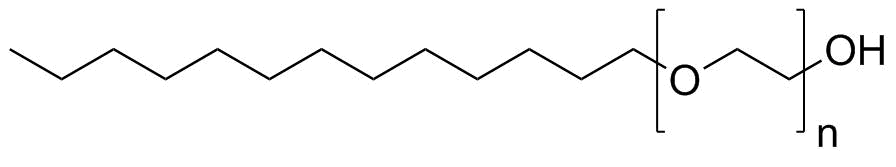 |
| CAS Registry Number | 9043-30-5 |
| EC Number | 500-027-2 |
| SMILES | CCCCCCCCCCCCOCCOCCOCCOCCOCCOCCOCCOCCOCCO |
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Isotridecanol ethoxylate is a nonionic surfactant produced by the ethoxylation of isotridecanol, a branched-chain C13 alcohol. It belongs to the class of alcohol ethoxylates, which are widely used in detergents, industrial cleaners, and emulsifying systems due to their effective surface-active properties and relatively good biodegradability compared with older alkylphenol-based surfactants. The parent alcohol, isotridecanol, is a synthetic branched primary alcohol typically derived from oligomerization of propylene followed by hydroformylation and hydrogenation. This process yields a mixture of highly branched C13 isomers rather than a single defined structure. The branching in the hydrocarbon chain reduces crystallinity and improves fluidity, which is advantageous in liquid surfactant formulations. Ethoxylation of isotridecanol is carried out by reacting the alcohol with ethylene oxide under alkaline catalysis. This introduces repeating ethylene oxide units (–CH2CH2O–)n onto the oxygen atom of the alcohol, forming a polyoxyethylene chain. The resulting product is not a single compound but a distribution of molecules with varying ethoxylation degrees, typically described by an average number of ethylene oxide units. The molecular structure consists of a hydrophobic branched C13 isotridecyl group attached to a hydrophilic polyoxyethylene chain. This amphiphilic configuration allows the molecule to act at interfaces between oil and water, reducing surface tension and stabilizing emulsions. The balance between hydrophobic and hydrophilic segments determines its hydrophilic–lipophilic balance (HLB), which is critical for its performance in different applications. In aqueous systems, isotridecanol ethoxylates form micelles once the concentration exceeds the critical micelle concentration. These micelles can solubilize oils, grease, and organic contaminants, making them effective detergents and emulsifiers. Because they are nonionic, they are relatively insensitive to water hardness and maintain performance in the presence of electrolytes. Isotridecanol ethoxylates are widely used in industrial and institutional cleaning formulations, including hard surface cleaners, degreasers, textile auxiliaries, and metal cleaning agents. They are also used in agricultural formulations as emulsifiers and dispersants for pesticides, as well as in emulsion polymerization processes in the chemical industry. One of their important characteristics is tunability: by adjusting the number of ethylene oxide units, manufacturers can tailor properties such as solubility, foaming behavior, wetting ability, and cloud point. Lower ethoxylation grades tend to be more lipophilic and better suited for emulsification of oils, while higher ethoxylation grades are more water-soluble and effective in aqueous cleaning systems. From a physicochemical perspective, the branched structure of isotridecanol contributes to lower viscosity and improved low-temperature performance compared with linear alcohol ethoxylates. The ethoxylate chain provides hydrogen bonding capability with water, while the alkyl chain interacts with hydrophobic substances, enabling efficient interfacial activity. Compared with older surfactant systems such as nonylphenol ethoxylates, isotridecanol ethoxylates are generally considered more environmentally acceptable due to improved biodegradation profiles and reduced formation of persistent degradation products. This has led to their increased use as replacement surfactants in many formulations. Overall, isotridecanol ethoxylate is a versatile nonionic surfactant derived from branched C13 alcohol and ethylene oxide. Its amphiphilic structure provides effective emulsifying, wetting, and detergency properties, making it widely used in industrial, institutional, and agricultural chemical formulations. References none |
| Market Analysis Reports |