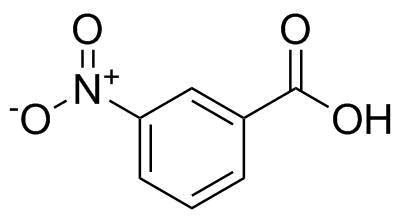
3-Nitrobenzoic acid is an aromatic carboxylic acid containing both a carboxyl group (–COOH) and a nitro group (–NO2) attached to a benzene ring in the meta position relative to each other. Its molecular formula is C7H5NO4. The presence of these two strongly electron-withdrawing functional groups gives the compound distinctive chemical reactivity and makes it a useful intermediate in organic synthesis.
The structure consists of a benzoic acid framework substituted at the 3-position by a nitro group. The carboxyl group is strongly polar and capable of forming hydrogen bonds, while the nitro group is a powerful electron-withdrawing substituent that influences both acidity and aromatic reactivity. Because of this substitution pattern, the compound exhibits increased acidity compared with unsubstituted benzoic acid, as the nitro group stabilizes the carboxylate anion through inductive effects.
The chemistry of nitrobenzoic acids became important during the development of aromatic nitration reactions in the 19th century, when electrophilic aromatic substitution was extensively studied. The controlled nitration of benzoic acid derivatives allowed chemists to explore directing effects of substituents on aromatic rings. It was established that the carboxyl group is a meta-directing deactivator, which explains why nitration of benzoic acid preferentially yields the 3-nitro isomer under appropriate conditions.
Industrial and laboratory preparation of 3-nitrobenzoic acid is typically achieved by nitration of benzoic acid using a mixture of nitric acid and sulfuric acid. The reaction proceeds through electrophilic aromatic substitution, where the nitronium ion (NO2+) acts as the electrophile. The carboxyl group directs substitution to the meta position, resulting in selective formation of the 3-nitro derivative. Reaction conditions are carefully controlled to minimize formation of other isomers or over-nitrated byproducts.
3-Nitrobenzoic acid is widely used as an intermediate in organic synthesis. The nitro group can be reduced to an amino group, yielding 3-aminobenzoic acid, which serves as a precursor for dyes, pharmaceuticals, and functional materials. This reduction can be carried out using catalytic hydrogenation or chemical reducing agents such as iron in acidic media. The resulting amino derivative can undergo further functionalization, including diazotization and coupling reactions.
The compound is also used in the preparation of more complex aromatic systems. The carboxyl group allows for esterification, amidation, and conversion to acid chlorides, enabling incorporation into polymers and small-molecule derivatives. Meanwhile, the nitro group serves as a versatile functional handle for subsequent transformations in synthetic chemistry.
In addition to its role as a synthetic intermediate, 3-nitrobenzoic acid has been used in academic research to study electronic effects in substituted aromatic systems. Its well-defined electronic structure makes it a useful model compound for investigating substituent effects on acidity, reactivity, and spectroscopic properties.
Overall, 3-nitrobenzoic acid is a substituted aromatic carboxylic acid characterized by the presence of a meta-positioned nitro group that significantly influences its chemical behavior. Its discovery and use are closely tied to the development of aromatic substitution chemistry, and it remains an important intermediate in the synthesis of dyes, pharmaceuticals, and other functional organic compounds.
References
2026. Effect of Temperature on Sodium Perchlorate Radiolysis under Extraterrestrial Conditions. Solar System Research.
DOI: 10.1134/s0038094625601069
2025. A benzylidene-amine scaffold as a colourimetric sensor for picric acid: computational studies and real-time applications using matchstick head powder. BMC Chemistry.
DOI: 10.1186/s13065-025-01670-4
2025. Polymorph Analysis of ALZ-801 (Valiltramiprosate), a Valine-Conjugated Oral Prodrug of Tramiprosate in Late-Stage Clinical Development for Alzheimer’s Disease. Journal of Chemical Crystallography.
DOI: 10.1007/s10870-025-01047-4
|



































































 GHS07 Warning Details
GHS07 Warning Details