Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Eastar Chemical Corporation | USA | |||
|---|---|---|---|---|
 | www.eastarchem.com | |||
 | +1 800-898-2436 | |||
 | +1 (877) 898-2436 | |||
 | info@eastarchem.com | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Bydone (Shanghai) Biochemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bydone-chem.com | |||
 | +86 15826115498 | |||
 | sales@bydonechem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2020 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Ningbo Wingoo Semiconductor Core Material Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.wingooxc.com | |||
 | +86 18842662104 | |||
 | zoulin@wingooxc.com | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Porton Corporation | China | |||
|---|---|---|---|---|
 | www.porton.cn | |||
 | +86 (23) 6763-5888 | |||
 | +86 (23) 6703-8513 / 6763-5889 | |||
 | porton@porton.cn | |||
| Chemical manufacturer since 2005 | ||||
| Classification | Organic raw materials >> Hydrocarbon compounds and their derivatives >> Cyclic hydrocarbon |
|---|---|
| Name | Benzocyclobutene |
| Synonyms | Bicyclo[4.2.0]octa-1,3,5-triene |
| Molecular Structure | 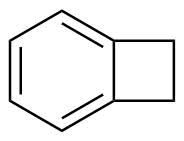 |
| Molecular Formula | C8H8 |
| Molecular Weight | 104.15 |
| CAS Registry Number | 694-87-1 |
| EC Number | 633-274-0 |
| SMILES | C1CC2=CC=CC=C21 |
| Density | 1.0$+/-$0.1 g/cm3 Calc.*, 0.957 g/mL (Expl.) |
|---|---|
| Boiling point | 164.3$+/-$10.0 $degree$C 760 mmHg (Calc.)*, 150.6 $degree$C (Expl.) |
| Flash point | 40.1$+/-$7.1 $degree$C (Calc.)* |
| Index of refraction | 1.586 (Calc.)*, 1.541 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS02;GHS07 WarningGHS02; Details GHS02;GHS07 WarningGHS02; Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H332-H412 Details | ||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P261-P271-P273-P280-P303+P361+P353-P304+P340-P317-P370+P378-P403+P235-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| Transport Information | UN 3295 | ||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Benzocyclobutene is a bicyclic organic compound consisting of a benzene ring fused to a cyclobutane ring. Its molecular formula is C8H8. The structure is characterized by a highly strained four-membered cyclobutene ring fused directly to an aromatic benzene ring, creating a rigid and reactive fused-ring system. The key structural feature of benzocyclobutene is the cyclobutene moiety, which contains significant ring strain due to the deviation of bond angles from the ideal tetrahedral geometry. This strain makes the compound chemically reactive under thermal or catalytic conditions. The benzene ring, in contrast, provides aromatic stabilization, and the fusion of these two rings leads to unique electronic and reactive properties. One of the most important characteristics of benzocyclobutene is its ability to undergo thermal ring-opening reactions. Upon heating, the cyclobutene ring can open to form an o-quinodimethane intermediate. This intermediate is highly reactive and can participate in pericyclic reactions such as Diels–Alder cycloadditions. This thermally induced transformation is reversible in some systems and is a key feature in polymer and materials chemistry. The discovery and study of benzocyclobutene derivatives became particularly important in the field of high-performance polymers and electronic materials. The thermal ring-opening behavior allows benzocyclobutene-containing monomers to act as latent reactive species, enabling controlled crosslinking reactions upon heating. This property is used in the design of thermosetting resins and low-dielectric materials for microelectronics. Benzocyclobutene itself can be synthesized through intramolecular cyclization reactions of appropriately substituted aromatic precursors. One common synthetic approach involves formation of the cyclobutene ring via photochemical or thermal rearrangements of styrene derivatives or related intermediates. The synthesis requires careful control due to the inherent strain and reactivity of the four-membered ring. In materials chemistry, benzocyclobutene-based monomers and polymers are widely studied for applications in microelectronics, particularly as low dielectric constant materials in integrated circuits. Upon thermal activation, the benzocyclobutene groups open to form reactive o-quinodimethane species, which can undergo further coupling reactions to form crosslinked networks. These networks exhibit high thermal stability, chemical resistance, and favorable electrical insulating properties. The reactivity of benzocyclobutene is governed by the balance between aromatic stabilization and cyclobutene ring strain. The strain energy stored in the four-membered ring is released upon ring opening, making the transformation thermodynamically favorable at elevated temperatures. This makes benzocyclobutene a useful “thermally latent” functional group in synthetic chemistry. Beyond polymer applications, benzocyclobutene derivatives are also used in organic synthesis as reactive intermediates for constructing complex polycyclic structures. The o-quinodimethane intermediate generated upon ring opening can participate in cycloaddition reactions that build larger fused-ring systems. Overall, benzocyclobutene is a strained bicyclic aromatic compound with a fused benzene–cyclobutene structure. Its unique thermal ring-opening behavior and ability to generate highly reactive intermediates make it an important building block in polymer chemistry, materials science, and synthetic organic chemistry, particularly in applications requiring controlled thermal activation and crosslinking. References 2026. Activation of alcohols as sulfonium salts in the photocatalytic hetero-difunctionalization of alkenes. Nature Chemistry. DOI: 10.1038/s41557-025-02003-7 2025. Design a compact square-ring antenna using a thin film of HTS materials. Journal of Computational Electronics. DOI: 10.1007/s10825-025-02369-4 2025. Catalyst Supports. Springer Series in Materials Science. DOI: 10.1007/978-3-031-85304-3_6 |
| Market Analysis Reports |